P2: Dipeptidyl-peptidases in cell signalling
Dipeptidyl-peptidases in signalling and stress responses (PI Ruth Geiss-Friedlander)
Dipeptidyl peptidases of the S9B family include four active members: DPPIV, DPP8, DPP9 and FAP. Due to their rare ability to cleave post-proline, these proteases often play key regulatory roles, for example in inflammasome activity, in B Cell receptor mediated signaling and cytokine processing. The underlying mechanisms are in most cases not well understood. DPP8 and DPP9 present the two intracellular members of this family, localizing to the cytoplasm, the nucleus and are associated with the plasma membrane.
ProtPath offers two topics for doctoral work on DPPs:
1) DPP8 and DPP9 in B cell signalling
Syk is a tyrosine kinase, playing a central role in the B cell receptor (BCR) mediated signalling. DPP9 cleaves the N-terminus of Syk to expose and N-degron, resulting in Syk degradation by the N-end rule pathway. By targeting Syk for degradation, DPP9 regulates the strength and the duration of Syk signalling.
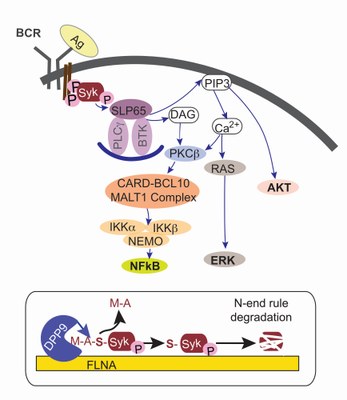
Syk in BCR (upper panel): Binding of an antigen to the BCR initiates the phosphorylation of the tyrosine kinase Syk. Phosphorylated Syk is active and initiates a cascade of phosphorylation events thereby regulating cell fate.
Syk is a DPP9 substrate (lower panel): DPP9 cleaves the N-terminus of activated Syk, targeting Syk for degradation by the N-Degron (N-end rule) pathway.
Reference: Justa-Schuch, D., Silva-Garcia, M., Pilla, E., Engelke, M., Kilisch, M., Lenz, C., Möller, U., Nakamura, F., Urlaub, H., Geiss-Friedlander, R. (2016). DPP9 is a novel component of the N-end rule pathway targeting the tyrosine kinase Syk. eLife 5, e16370. doi:10.7554/eLife.16370
The following questions will be addressed in this doctoral project:
Is the DPP8/9 substrate repertoire altered upon BCR-stimulation?
2. How are DPP8 and DPP9 regulated (how do they meet their substrates)?
3. What are the molecular and functional consequces of the cleavage event?
These questions will be addressed by combining Protein N-terminomic studies and Interactome studies. Validation of substrates and interacting partners will be performed in vitro and in cell-based assays, followed by biological readouts.
2) DPP9 in the Unfolded Protein Response (UPR)
The UPR is a stress response triggered by the accumulation of misfolded proteins in the endoplasmic reticulum (ER). To reduce the load of proteins within the ER the UPR increases the expression of proteins involved in protein folding and proteins involved in the degradation of ER proteins (ER associated protein degradation), in addition to attenuation of mRNA translation. Defects in the UPR are linked to neurodegeneration diseases, cancer, and inflammation.
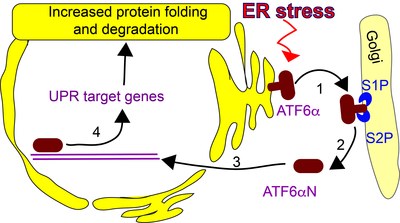
ATF6 in the UPR: (1) ATF6 translocates to the golgi upon ER stress, where (2) it is sequentially cleaved by two proteases: S1P and S2P. (3) The liberated N-terminal ATF6 fragment (ATF6N) is a transcription factor. ATF6N enters the nucleus and (4) upregulates the expression of target genes.
In a screen for DPP9 substrates we identified ATF6, an ER stress sensor and transducer of the unfolded protein response (UPR). In this project the role of DPP9 in the UPR will be examined by studying how does DPP9 affect ATF6 activity on the molecular level? How is processing of ATF6a by DPP9 regulated? What are the cellular consequences?
These questions will be addressed by combining molecular biology, biochemical and experiemnts in mammalian cell-culture.
Reference: Zhang, H; Maqsudi, S., Rainczuk, A., Duffield, N; Lawrence, J., Keane, FM., Justa-Schuch, D., Geiss-Friedlander, R., Gorrell, MD., Stephens, AN. Identification of Novel Dipeptidyl Peptidase 9 Substrates by 2D DIGE. 2015 FEBS Pubmed326175140 doi: 10.1111/febs.13371.

