P6: Proteases in autophagy initiation
The role of Atg4 protease in autophagy initiation and regulation (PI Claudine Kraft)
Autophagy is a lysosomal degradation pathway in which the cell digests its own components. During autophagy, cytoplasmic material is enclosed within autophagosomes and transported to the lytic compartment (the vacuole i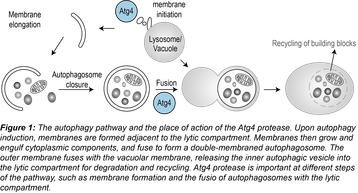 n yeast, the lysosome in mammals) for degradation and recycling (Figure 1). Autophagy is emerging as a central biological pathway that promotes health and longevity. On the other hand, defects in autophagy have been associated with several human diseases, including neurodegenerative disorders and cancer. Modulation of autophagic flux is a new field being explored in the search for novel therapies for various diseases. Our goal is to dissect autophagy at a molecular level, so that we can understand its dysfunction in disease and unleash its potential as a therapeutic target.
n yeast, the lysosome in mammals) for degradation and recycling (Figure 1). Autophagy is emerging as a central biological pathway that promotes health and longevity. On the other hand, defects in autophagy have been associated with several human diseases, including neurodegenerative disorders and cancer. Modulation of autophagic flux is a new field being explored in the search for novel therapies for various diseases. Our goal is to dissect autophagy at a molecular level, so that we can understand its dysfunction in disease and unleash its potential as a therapeutic target.
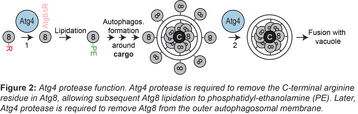 The autophagic pathway and its molecular machinery is highly conserved from yeast to mammals. Autophagy is initiated at a perivacuolar site, where autophagic membranes form. A key step during autophagosome biogenesis is the conjugation of the ubiquitin-like protein Atg8 to phosphatidylethanolamine (PE), which is a prerequisite for autophagic membrane formation and elongation. Atg4 is a cysteine protease essential for autophagy and plays two critical roles. First, Atg4 primes Atg8 by proteolytically removing its C-terminal Arginine (R) residue, allowing Atg8 to be lipidated to PE (Figure 2, step 1). Later, Atg4 removes Atg8 from the completed autophagosome, which has been suggested to regulate timely fusion with vacuoles (Figure 2, step 2). We have recently contributed to the finding that Atg4 proteolytic activity is under the control of the Atg1 kinase (Sanchez-Wandelmer et al., 2017) and to the understanding of how Atg8 and Atg4 interact (Abreu et al., 2017) in collaboration with the Reggiori group (UMC Groningen, NL).
The autophagic pathway and its molecular machinery is highly conserved from yeast to mammals. Autophagy is initiated at a perivacuolar site, where autophagic membranes form. A key step during autophagosome biogenesis is the conjugation of the ubiquitin-like protein Atg8 to phosphatidylethanolamine (PE), which is a prerequisite for autophagic membrane formation and elongation. Atg4 is a cysteine protease essential for autophagy and plays two critical roles. First, Atg4 primes Atg8 by proteolytically removing its C-terminal Arginine (R) residue, allowing Atg8 to be lipidated to PE (Figure 2, step 1). Later, Atg4 removes Atg8 from the completed autophagosome, which has been suggested to regulate timely fusion with vacuoles (Figure 2, step 2). We have recently contributed to the finding that Atg4 proteolytic activity is under the control of the Atg1 kinase (Sanchez-Wandelmer et al., 2017) and to the understanding of how Atg8 and Atg4 interact (Abreu et al., 2017) in collaboration with the Reggiori group (UMC Groningen, NL).
In this graduate school, we aim at understanding the molecular role of Atg4 protease in autophagosome biogenesis and fusion with the vacuole.
Thesis topic 1: The role of Atg4 protease in timely autophagosome-vacuole fusion
Thesis topic 2: Atg4 protease interaction network in yeast and mammals
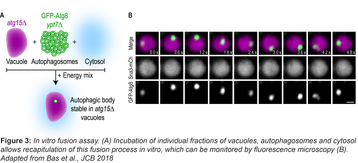 The core autophagic machinery is highly conserved from yeast to humans. Therefore, we use both mammalian cell culture and budding yeast as model systems to discover the basic principles of autophagy. Our approaches range from classical reconstitutions, using recombinant proteins and purified membranes, to reconstitutions using cell fractions (Figure 3) and synthetic in vivo approaches (Torggler et al., 2016). We also analyze intact cells.
The core autophagic machinery is highly conserved from yeast to humans. Therefore, we use both mammalian cell culture and budding yeast as model systems to discover the basic principles of autophagy. Our approaches range from classical reconstitutions, using recombinant proteins and purified membranes, to reconstitutions using cell fractions (Figure 3) and synthetic in vivo approaches (Torggler et al., 2016). We also analyze intact cells.
References
Abreu S, Kriegenburg F, Gomez-Sanchez R, Mari M, Sanchez-Wandelmer J, Rasmussen M, Soares Guimaraes R, Zens B, Schuschnig M, Hardenberg R, Peter M, Johansen T, Kraft C, Martens S and Reggiori F (2017). A conserved LIR motif mediates Atg4 association to autophagosomal membranes and Atg8 deconjugation. EMBO Reports 18: 765-780. DOI: 10.15252/embr.201643146
Bas L, Papinski D, Licheva M, Torggler R, Rohringer S, Schuschnig M, and Kraft C (2018). Reconstitution reveals Ykt6 as the autophagosomal SNARE in autophagosome-vacuole fusion. Journal of Cell Biology 217 (10): 3656-3669. DOI: 10.1083/jcb.201804028
Sánchez-Wandelmer J, Kriegenburg F, Rohringer S, Schuschnig M, Gomez-Sanchez R, Zens B, Abreu S, Hardenberg R, Hollenstein D, Gao J, Ungermann C, Martens S, Kraft C and Reggiori F (2017). Atg4 proteolytic activity can be inhibited by Atg1 phosphorylation. Nature Communications 8: 295. DOI: 10.1038/s41467-017-00302-3
Torggler R, Papinski D, Brach T, Bas L, Schuschnig M., Pfaffenwimmer T., Rohringer S, Matzhold T, Schweida D, Brezovich A and Kraft C (2016). Two Independent Pathways within Selective Autophagy Converge to Activate Atg1 Kinase at the Vacuole. Molecular Cell 64: 221-235. DOI:10.1016/j.molcel.2016.09.008

