P8: Orphan proteases of mitochondria
Mitochondrial Proteases: Novel players and regulatory mechanisms (PI Chris Meisinger)
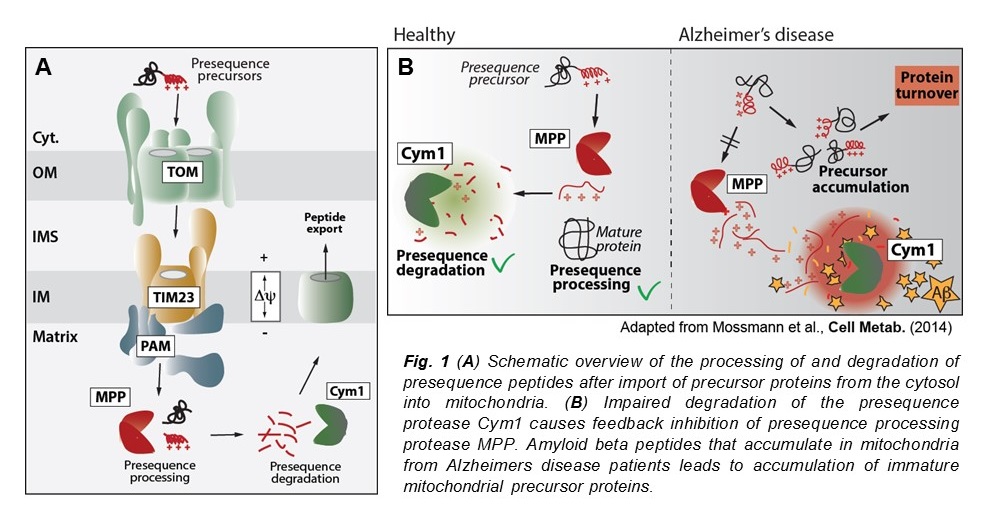
Our research focuses on the discovery of novel mitochondrial proteases and study their role, particularly of processing of imported precursor proteins and degradation of presequence peptides. We have identified several novel mitochondrial proteases (1,2) and also found that impaired presequence peptide degradation plays an important role in the onset of Alzheimers disease (3). Impaired presequence processing can also cause very early steps in the mitochondrial unfolded protein pathway mtUPR (4).
ProtPath offers several topics for doctoral work on the the analysis of novel (orphan) mitochondrial proteases and their role in health and disease:
By using various proteomics approaches including a systematic profiling of the exact suborganellar protein composition and localization (Landscape of submitochondrial protein distribution) we could identify several novel proteases located in the mitochondrial matrix of the yeast S. cerevisiae (2). This includes several metallo-aminopeptidases with no annotated function yet and the cysteine-aminopeptidase Lap3. We could confirm the matrix localization of these enzymes biochemically, however, their functions remain entirely unknown. Most of these proteases are highly homolog to human genes encoding putative proteases. Our project aims to elucidate the functional role of these novel mitochondrial orphan proteases. This will include systematic profiling for sustrate proteins using N-terminomics approaches and characterize mutant variants in the model organisms bakers yeast and human. Functional studies will also include in vitro and in organello assays to profile processing or degradation of identified substrates. We also aim to decipher regulatory signalling networks that might regulate protease activities as response to changing cellular needs. This will include profiling and analysis of posttranslational modification (e.g. by reversible phosphorylation) (5).
References
1. Vögtle, F.N., Wortelkamp, S., Zahedi, R.P., Becker, D., Leidhold, C., Gevaert, K., Kellermann, J., Voos, W., Sickmann, A., Pfanner, N., and Meisinger, C. (2009) Global analysis of the mitochondrial N-Proteome identifies a processing peptidase critical for protein stability. Cell 139, 428-439
2. Vögtle, F.N., Burkhart, J.M., Gonczarowska-Jorge, H., Kücükköse, C., Taskin, A.A., Kopczynski, D., Ahrends, R., Mossmann, D., Sickmann, A., Zahedi, R.P., and Meisinger, C. (2017) Landscape of submitochondrial protein distribution. Nat. Commun. 8, 290
3. Mossmann, D., Vögtle, F.N., Taskin, A.A., Teixeira, P.F., Ring, J., Burkhart, J.M., Burger, N., Pinho, C.M., Tadic, J., Loreth, D., Graff, C., Metzger, F., Sickmann, A., Kretz, O., Wiedemann, N., Zahedi, R.P., Madeo, F., Glaser E., and Meisinger, C. (2014) Amyloid-b peptide induces mitochondrial dysfunction by inhibition of preprotein maturation. Cell Metab. 20, 662-669
4. Poveda-Huertes, D., Matic, S., Marada, A., Habernig, L., Licheva, M., Myketin, L., Gilsbach, R., Tosal-Castano, S., Papinski, D., Mulica, P., Kretz, O., Kücükkose, C., Taskin A.A., Hein, L., Kraft, C., Büttner, S., Meisinger, C., and Vögtle, F.N. (2019). An early mtUPR: Redistribution of the nuclear transcription factor Rox1 into mitochondria protects against intramitochondrial proteotoxic aggregates. Mol. Cell 77, 180-188.
5. Schmidt, O., Harbauer, A.B., Rao, S., Eyrich, B., Zahedi, R.P., Stojanovski, D., Schönfisch, B., Guiard, B., Sickmann, A., Pfanner, N., and Meisinger, C. (2011) Regulation of mitochondrial protein import by cytosolic kinases. Cell 144, 227-239

